- Home
- Details
- Registry
- RSVP
- Blog
- Blog
- Minigame lobby secrets minecraft xbox 360
- C1s c-n xps peak table
- Melodyne 3 64 bit
- Ekla chalo re translation
- Kutools for excel change case
- Linkin park hybrid theory album artwork
- Matlab sort
- Free iso burner to usb flash drive
- Tutorial surfer 9
- Iptv m3u playlist android
- I wanna be adored stone roses guitar rig presets
- Infectonator 3 apocalypse hacked
- Ti 84 emulator tom
- Fl studio 20 reg key only
- Best diff tool for ubunutr
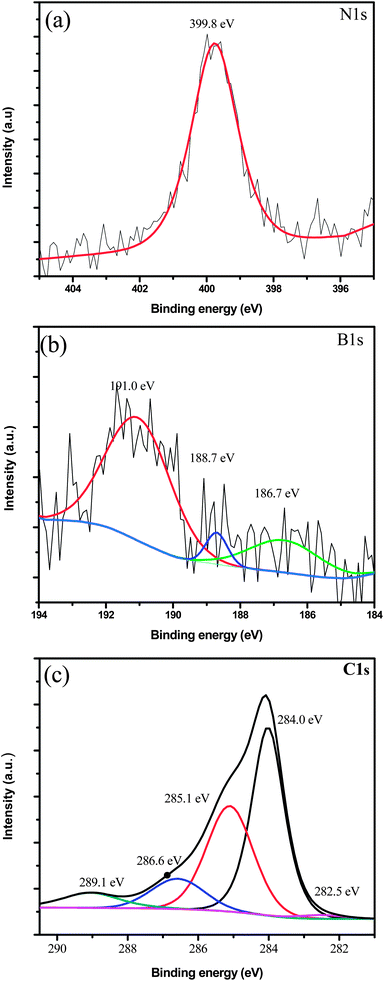
C1s regions for polymers can often have acquisition times of Different polymers have different degradation rates.Some polymers are sensitive to prolonged X-ray exposure.Particularly useful in cases where C1s XPS data cannot easily distinguish between.Acquisition of valence band can be useful for “fingerprint” identification of polymers.Polymers Binding energies of common chemical states: Inelastic scatter of electrons through the adventitious carbon layer may cause a largeĬhange in background shape when compared with the spectrum from a clean sample.For example, the Na1s peak (binding energy=1071eV) may be weaker thanĮxpected, or completely disappear, but the Na KLL peak (binding energy=497eV).The intensity of higher binding energy peaks may be reduced relative to their.Quantification may be inaccurate when using peaks of significantly different energy.On native oxide of aluminium appears at ~286eV).Ī few nanometers of adventitious carbon on a sample can dramatically affect the survey Not always a valid charge reference value (e.g.The C-C component may be set to a binding energy of 284.8eV, by default.C1s spectrum for contamination typically has C-C, C-O-C and O-C=O components.Prevent/minimize chemical damage to the underlying material.Īdventitious carbon contamination is commonly used as a charge reference for XPS spectra. Use lowest energy argon beam available (e.g.Adventitious carbon may be removed by argon sputtering.(Typically with a layer thickness of 1-2nm.)

Most samples that have been exposed to the atmosphere will have a detectable quantity ofĪdventitious carbon contamination.Carbonates and Carbides)Īdventitious Carbon Contamination Binding energies of common chemical states:Ĭharge referenced to adventitious C1s, C-C peak at 284.8eV Experimental Information
- Home
- Details
- Registry
- RSVP
- Blog
- Blog
- Minigame lobby secrets minecraft xbox 360
- C1s c-n xps peak table
- Melodyne 3 64 bit
- Ekla chalo re translation
- Kutools for excel change case
- Linkin park hybrid theory album artwork
- Matlab sort
- Free iso burner to usb flash drive
- Tutorial surfer 9
- Iptv m3u playlist android
- I wanna be adored stone roses guitar rig presets
- Infectonator 3 apocalypse hacked
- Ti 84 emulator tom
- Fl studio 20 reg key only
- Best diff tool for ubunutr
